We put forward and schematic approach of the different anatomic structures conforming the visual pathway and most relevant pathologic entities affecting them.
The optic nerve is the second cranial nerve but technically forms part of the central nervous system, as it contains the same tissue components as the brain. It can be divided into intraocular, intraorbital, canalicular and intracranial segments.
The intraorbital optic nerve has a length of about 4-4.5cm, running posteromedially to the cone formed by the rectus muscles, and enters into the optic canal.The entire intraorbital nerve is embedded in meningeal and arachnoidal sheaths in continuation with the respective intracranial layers, corresponding the outtermost sheath to dura matter, and the inner sheaths to arachnoid layers. Inbetween the intermediate and the inner layers of the arachnoid there is a tubular-shaped subarachnoid space, communicating with the intracranial subarachnoid space, and which can be widened proximally not implying pathology. Passing the optic canal, this tubular subarachnoid space narrows in normal conditions.
The intracanalicular segment has 5mm of lenght, and the intracranial segment 10mm. Internal carotid arteries lie laterally and inferiorly to the intracranial optic nerve, arising the ophtalmic arteries from the anteromedial carotid syphon, entering the orbita into the subarachnoid space through the optic canal, infero-laterally to the optic nerve, and piercing the dura into the orbital side of the canalicular segment of the optic nerve. The A1 segments of the anterior cerebral arteries cross above the prechiasmal optic nerve.
Lesions affecting the retina or the optic nerve will result in some degree of ipsilateral visual field defect. Optic nerve pathology can be divided into intrinsic and extrinsic lesions.
Intrinsic lesions include optic neuritis (ON) and ischemic optic neuropathy (ION).
· ON cardinal symptoms are usually central visual field loss (although it may result in a large variety of visual field loss) and pain in the affected eye.
· ION is a collective name for a number of ischemic syndromes affecting the optic nerve.
There is a large range of extrinsic lesions that may result in compressive optic neuropathy (CON). Clinical symptomatology includes optic disc swelling, proptosis, ophthalmoplegia, congestion and visual lose, usually with slow onset, which might be the clinical clue to differentiate CON from ON or ION. MR is the gold standard diagnostic tool, although CT also plays a role in some circumstances.
The differential diagnosis includes:
· Thyroid-related ophthalmopathy.
· Optic nerve tumors: optic nerve glioma, optic nerve sheath meningioma, lymphoproliferative syndromes.
· Optic nerve inflammatory disease: sarcoidosis, orbital pseudotumor.
· Intraorbital expansive lesions:
Ø Vascular: Cavernous hemangioma, cystic lymphangioma, orbital venous varix.
Ø Tumoral: lymphoproliferative syndromes
Ø Traumatic: direct or indirect optic nerve lesion.
Ø Infectious: postseptal orbital cellulitis with subperiostic abscess.
· Intracranial hypertension.
· Glaucoma.
The x-shaped optic chiasm forms from the confluence in the midline of the intracranial segments of the optic nerves in the chiasmatic sulcus, superoanterior to the ridge of the dorsum sellae. The medial fibers (temporal visual field) deccusate to the contralateral site, whereas the lateral fibers do not. The posterior margin of the chiasm partially forms the anterior floor of the third ventricle. Posterior to the chiasm,the tuber cinereum and the pituitary stalk extends inferiorly.
The range of lesions affecting the chiasm makes MR the preferred imaging technique. Lesions in the optic chiasm regions can cause a variety of visual symptoms, being the bitemporal hemianopia the most characteristic defect and is due to central chiasm affectation from extrinsic compressive lesions. It is worth noting that infiltrating or inflammatory lesions do not tend to produce stereotypical visual field defects and visual loss may not correlate to the extent or location of the lesion.
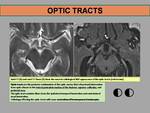
Optic tracts are the anatomic structures that continue backwards the optic visual pathway, emerging dorsolaterally from the optic chiasm. They cross posterior to the anterior perforated substance, and anterior to the lateral tuber cinereum and to the lateral perforated substance. Dorsolaterally optic tracts lie above the medial temporal lobes and surround the crus cerebri. Most fibers of the optic tracts terminate in the lateral geniculate nucleus of the thalami, but there are several small divisions ending in the superior colliculus, pretectal area, accesory optic tract nucleus and hypotalamic suprachiasmatic nucleus.
Lesions posterior to the optic chiasm result in homonymous visual field defects without loss of of acuity. Generally neoplastic lesions produce a gradual onset of symptoms in contrast to the sudden onset with vascular lesions.
Homonymous hemianopia due to optic tract or LGN lesion are rare, representing altogether approximately the 10% of all. Optic tracts are susceptible to lesions arising from the optic chiasm but also from medial temporal lobes and midbrain. LGN lesions usually are due to infarction of anterior or lateral choroid arteries. Lateral and medial portions of the LGN represent the superior and inferior hemifields. Anterior choroid artery infarction (irrigation of both lateral and medial portions) will result in a peripheral wedge-shaped homonymous hemianopia, and lateral choroid artery infarction results in a homonymous horizontal quadrantanopia (irrigation of the hilum of the LNG).
Optic radiations consist of the different divisions of fibers that connect the lateral geniculate nucleus and other minor nuclei to the visual cortex. Half of the fibers originating in the lateral geniculate nucleus run directly backward lateral to the more superior aspect of the lateral ventricle within the inferior parietal lobe. The other half of the fibers describes a large forward directed loop to the temporal lobe, above and lateral to the temporal horn, called Meyer's loop, and the curve backwards lateral to the temporal and occipital horn of the lateral ventricle.
Lesions affecting specific groups of fibers will develop different visual field defects.
· Lesions in the inferior temporal components: wedge-shaped superior contralateral quadrantanopia.
· Lesions in the superior parietal components: Inferior contralateral quadrantanopia.
· Large lesions affecting both components: homonymous hemianopia with macular splitting.
The primary visual cortex is located in the wall of the calcarine fissures, on the medial surface of the occipital lobes. The folding of the calcarine fissure indicates the horizontal meridian, being the lower contralateral visual field represented above the horizontal meridian, whereas the upper contralateral visual field is represented below it. The most caudal part, extending to the pole represents the fovea and the volume of the tissue is relatively large compared to the peripheral field area, that occupies the rostral portions.
Lesions affecting the occipital lobe account for nearly 50% of homonymous visual field defects with infarction, secondary to MCA or PCA, accounting for nearly 75% of the overall. Lesions of the occipital pole produce contralateral large homonymous scotomas that are extremely congruous, and more anterior lesions progressively involve the peripheral vision and again are highly congruous.
Bilateral occipital lobe lesions occurring simultaneously, or more frequently, sequentially develop variable degrees of bilateral homonymous hemianopia with or without macular sparing. If there is complete damage of the primary visual cortex, it results in cortical blindness.
primary visual cortex, it results in cortical blindness.